Concrete looks simple: sand, stone, cement, water. Up close, though, it is a busy chemical workshop where minerals dissolve, recrystallize, and weave a microscopic fabric that supports highways, skyscrapers, and homes. Understanding how concrete gains strength starts at the moment water meets cement and continues for years as reactions evolve and microstructure tightens.
A quick tour of the raw materials
Portland cement, the most common binder, is not a single mineral but a manufactured blend of calcium silicates, aluminates, and ferrites with a bit of gypsum added. Each of these clinker phases reacts differently with water: some are energetic and fast, others are slow but important for long-term strength. The balance built into a cement’s chemistry controls setting times, heat release, and how the paste microstructure forms.
Aggregates — sand and gravel — are inert in the short term but vital as the skeleton that the cement paste binds together. Their size, shape, and mineralogy affect packing density and permeability. Water is the trigger: it activates chemical reactions and also sets the initial workability that determines how well the mix can be placed and consolidated.
Supplementary cementitious materials (SCMs) such as fly ash, slag, and silica fume often join the mix to modify hydration. These materials participate in slower, pozzolanic or latent hydraulic reactions that consume certain hydration products and create additional binding phases. Designers use SCMs to tune cost, durability, and carbon footprint, but they also change how and when strength develops.
What really happens when water meets cement
The core of strength development is hydration — a set of chemical reactions between cement phases and water that produce new solids and soluble ions. The dominant product responsible for binding is calcium silicate hydrate, commonly abbreviated C-S-H. C-S-H is not a single crystal but a gel-like, poorly ordered solid that fills space and glues aggregate particles together.
When tricalcium silicate (C3S) and dicalcium silicate (C2S) react with water, they dissolve and re-precipitate as C-S-H plus calcium hydroxide (often called portlandite, CH). The simplified reactions look like this in cement chemistry shorthand: C3S + water → C-S-H + CH, and C2S + water → C-S-H + CH. The C-S-H is the “glue” while CH is a crystalline byproduct with little binding value.
Aluminate phases (mainly C3A) react rapidly with water in the presence of gypsum to form ettringite, a needle-like mineral, which governs early setting and can later transform to monosulfate as sulfate is depleted. The ferrite phase (C4AF) reacts too but contributes less to strength. Overall, these reactions reduce porosity and build a solid network, which is why strength increases over time.
Microstructure: the architecture of strength
Strength isn’t just about chemical products; it’s about how those products arrange themselves. C-S-H forms a pervasive, nanoscopic network of gel pores and sheets that bridge particles. The density and connectivity of this C-S-H network determine stiffness and compressive strength more than any single mineral percentage.
Two types of pores control the story: gel pores within C-S-H and capillary pores left where water once resided. Gel pores are tiny and contribute little to permeability, while capillary pores are larger and create weakness. As hydration proceeds, capillary pores shrink because more solid fills the voids; this densification increases strength and reduces permeability.
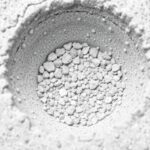
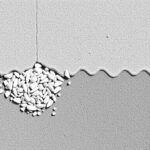
Microscopic images show C-S-H as a web of irregular particles and platelets that wrap around unhydrated clinker and aggregate grains. Where this web is continuous and well-bonded to aggregates, concrete resists compressive loads effectively. Where porosity or weak interfaces persist, cracks begin at lower stresses.
Stages of hydration and strength development
Hydration unfolds in recognizable stages. An initial dissolution period lasts minutes to hours, during which the paste is highly workable. This is followed by a dormant period that gives working time, then an acceleration stage when setting begins and early strength rises quickly. Finally, a deceleration stage spans days to years as the reactions slow and long-term strength accumulates.
Early strength (first 1–7 days) is dominated by C3S hydration and the formation of ettringite and initial C-S-H. C3S reacts relatively fast, releasing heat and building early stiffness. Later strength (beyond 7 days and especially past 28 days) benefits from slower C2S hydration and pozzolanic reactions that produce additional C-S-H and fill pores.
Because the rate of strength gain changes with time, engineers specify ages like 7 and 28 days for tests. Twenty-eight days became a conventional benchmark because it balances practical construction schedules with meaningful maturity of hydration, but many mixes continue to gain strength long after that point.
Water-to-cement ratio: the master variable
One of the simplest, most powerful rules in concrete is that lower water-to-cement (w/c) ratio produces higher strength. Less water means fewer and smaller capillary pores once hydration consumes the available water, so the hardened paste is denser. Abrams’ law summarizes this inverse relationship and remains a practical design principle for mix proportioning.
Lower w/c raises strength but also hurts workability if not countered. That’s where chemical admixtures like superplasticizers come into play: they reduce the need for extra water while maintaining flow. There is a trade-off, however — if water is insufficient for complete hydration, internal curing strategies or added water-retaining agents may be required.
Typical w/c values vary by application: a structural element might use 0.40–0.50, while cast-in-place slabs sometimes use 0.45–0.55 for ease of placement. High-performance concretes push below 0.35, relying on admixtures and SCMs to maintain workability and ensure adequate hydration.
Heat of hydration and temperature effects
Hydration is exothermic; it releases heat that raises the temperature of the concrete. In small pours this heat dissipates quickly, but in mass concrete elements it can produce significant internal temperatures that create thermal gradients. Those gradients generate stresses and may lead to cracking if not managed.
Temperature accelerates chemical kinetics. Warmer concrete gains early strength faster because reactions proceed more quickly. However, very high curing temperatures can lead to coarser, less-ideal C-S-H structures that sometimes reduce long-term strength and durability. Engineers balance early strength needs with long-term performance by controlling curing temperature and rate.
Mass concrete placements use staged pours, low-heat cements, or cooling pipes to limit peak temperature. Precast plants often employ controlled heat curing — steam or hot water — to speed early production without sacrificing long-term properties, but experienced operators monitor both temperature and moisture carefully.
Admixtures: sculpting hydration and performance
Chemical admixtures are an engineer’s toolkit for tailoring hydration. Superplasticizers (high-range water reducers) disperse cement grains and increase flow without added water, enabling low w/c and high strength. Retarders slow the rate of hydration to lengthen workability, while accelerators speed early strength gain when time is tight.
Air-entraining agents create microscopic air bubbles that improve freeze-thaw durability but slightly reduce compressive strength because air voids replace solid material. Shrinkage-reducing admixtures, corrosion inhibitors, and specialty polymers further alter the fresh and hardened properties by affecting hydration chemistry or microstructure formation.
Using admixtures effectively requires understanding the interactions with cement composition and SCMs. For example, some superplasticizers can react differently with high-alkali cements or slag blends, so trial mixes and local experience are essential. I’ve supervised pours where switching to an alternative superplasticizer resolved severe stickiness within minutes — a reminder that chemistry in the mixing bowl is sensitive and practical testing matters.
Supplementary cementitious materials and their chemistry
SCMs extend and refine hydration chemistry. Fly ash and silica fume are primarily siliceous and react with CH in pozzolanic reactions to form additional C-S-H. Ground granulated blast furnace slag (GGBFS) behaves as a latent hydraulic material, reacting with water in the presence of activators to produce cementitious products similar to C-S-H.
Pozzolanic reactions are slower than primary hydration, so mixes with high SCM content often show slower early strength but superior long-term strength and reduced permeability. Silica fume is so fine and reactive that even small additions dramatically densify the microstructure, increasing strength and often improving resistance to chloride penetration.
Blends must be designed intentionally. A high-volume fly ash mix can reduce Portland cement content and improve durability, but on cold projects it may delay strength gain enough to affect formwork schedules. Knowing the trade-offs and matching SCM type and content to project constraints is central to successful design.
How microstructure ties to mechanical behavior
Compressive strength in concrete is intimately connected to porosity, pore-size distribution, and the quality of the transition zone between paste and aggregate. The aggregate-paste interface is often the weak link; a well-hydrated, dense transition zone improves load transfer and crack resistance. C-S-H that effectively bridges particles and binds to aggregate surfaces raises stiffness and peak strength.
Fracture in concrete tends to initiate at flaws — microcracks, pores, and imperfect interfaces. The more homogeneous and less porous the paste, the fewer stress concentrators exist to start crack growth. Strategies that refine pore structure, such as low w/c, pozzolanic consumption of CH, and proper curing, all contribute to delaying crack initiation and improving strength.
Tests like scanning electron microscopy reveal how hydration products occupy spaces and bind grains. Mechanical testing correlates observed microstructural improvements with higher compressive and tensile strength, confirming that chemistry and microstructure are not abstract concepts but practical levers for performance.
Durability implications of hydration products
Hydration products influence how concrete interacts with its environment. C-S-H reduces permeability, limiting the ingress of deleterious agents like chlorides and sulfates. Portlandite, however, is relatively soluble and can be consumed during aggressive exposures, which changes the chemical balance and may weaken protective capacities.
Carbonation — the reaction of CO2 with calcium hydroxide and C-S-H — lowers pH and can depassivate embedded steel, increasing corrosion risk. Concretes with dense microstructures and lower CH availability due to pozzolanic reactions are generally less susceptible to deep carbonation, although very low permeability can paradoxically slow CO2 diffusion and change local dynamics.
Sulfate attack involves external sulfates reacting with aluminate phases and forming expansive minerals like ettringite, which can crack paste. Proper sulfate resistance comes from limiting reactive aluminates, using low-permeability paste, and employing sulfate-resistant cements or SCMs where exposure dictates. Hydration chemistry is therefore central to long-term durability, not just early strength.
Alkali-silica reaction and chemical balances

Alkali-silica reaction (ASR) is a chemical swelling phenomenon where reactive silica in aggregates reacts with alkalis from cement to form an expansive gel. This gel absorbs water, swells, and cracks the concrete. The availability of alkalis, moisture, and reactive silica controls ASR potential, so hydration chemistry and mix selection play a preventive role.
SCMs like fly ash and slag can mitigate ASR by reducing the pore solution alkalinity and binding alkalis into the solid phases. Glassy or poorly crystalline aggregates are particularly vulnerable, and petrographic screening of aggregates remains a standard practice. The chemistry of hydration interacts with aggregate mineralogy to determine whether a mix will remain stable for decades.
In practice, design choices that limit alkali content, use low-alkali cement, or include adequate SCMs reduce ASR risk. I recall a renovation project where replacing reactive aggregate sources and increasing fly ash content prevented an otherwise likely durability failure, saving both time and money over the structure’s lifespan.
Maintaining moisture: curing and internal curing

Curing is the act of preserving moisture and appropriate temperature to allow hydration to continue. If the surface dries prematurely, hydration stalls near the surface, leaving a weak, porous layer that undermines durability and surface strength. Simple steps like wet coverings, curing compounds, or continuous water spray can dramatically improve outcomes.
Internal curing uses pre-wetted lightweight aggregates or superabsorbent polymers to supply water from within during hydration. This is especially useful in low w/c mixes where external water is insufficient for complete hydration throughout the paste. Internal curing helps reduce autogenous shrinkage and promotes more complete strength development.
In hot or windy conditions, curing is easy to neglect but costly in consequences. I’ve seen otherwise excellent mixes fail to develop expected strength on site because exterior winds and direct sun robbed the surface of moisture. Proper planning and attention to curing logistics are as important as mix design.
Testing and monitoring hydration in practice
Engineers use a range of tests to follow hydration and correlate it to strength. Compressive strength tests at specified ages remain the workhorse for acceptance, but laboratory calorimetry provides insight into the rate and heat of hydration. X-ray diffraction, thermogravimetric analysis, and scanning electron microscopy reveal phase compositions and microstructure.
Non-destructive methods such as ultrasonic pulse velocity and maturity methods estimate strength development in the field. Maturity functions, which combine time and temperature history, let contractors predict in-place strength without destructive sampling. These tools increase confidence in early stripping and load-bearing decisions.
Isothermal calorimetry in the lab is particularly useful when evaluating new cement blends, SCMs, or admixture interactions. It reveals whether a mix will set too quickly, generate too much heat in a mass pour, or retard unexpectedly. Such insights are practical and prevent costly surprises on the job site.
Design and construction implications
Understanding hydration chemistry informs concrete specification throughout design and construction. Engineers choose w/c, SCM content, and curing regimes based on structural requirements and exposure conditions. Durability targets — chloride ingress limits, freeze-thaw resistance, and sulfate exposure — translate directly into choices that shape hydration and microstructure.
Construction sequencing and temperature control matter as much as mix proportions. For mass pours, low-heat cement and thermal control avoid cracking; for rapid-turnaround precast, controlled steam curing accelerates early strength without sacrificing later performance when managed properly. Good communication between design, production, and field teams ensures the chemistry is matched to practical realities.
Another practical point: quality control during batching is non-negotiable. Slight variations in water content or admixture dosing can shift the hydration trajectory. Experienced field staff and robust testing regimes reduce variability and help concrete perform as intended.
Real-world examples and experience
At a precast plant where I consulted, switching from a standard mix to one with 8% silica fume and a lower w/c raised 28-day strengths and reduced surface scaling in freeze-thaw cycles. The change required tighter mixing controls and a brief redesign of the curing cycle, but the durability benefits proved worth the effort for bridge components destined for harsh environments.
On a separate highway project involving a large foundation pour, the contractor used chilled water and embedded cooling pipes to manage peak temperatures. Thermocouples showed internal temperatures hundreds of degrees lower than a similar uncontrolled pour nearby, and the cooler cure prevented early thermal cracking that would have led to expensive repairs.
These experiences underline a simple truth: chemical understanding matters at the jobsite. Small engineering choices informed by hydration chemistry — material selection, curing method, temperature control — have outsized effects on long-term strength and service life.
Failure modes tied to hydration mismanagement
Poorly hydrated concrete fails in predictable ways. Under-curing leaves a weak, porous surface prone to abrasion and infiltration. Excess water from over-watering on-site yields low density and low strength. Uncontrolled heat in mass pours creates thermal cracking that compromises both strength and durability.
Other failures come from mismatched chemistry: an accelerator that produces rapid early set can trap internal stresses if the mix is stiff, leading to cracking. Or an incompatible superplasticizer may cause slump loss or delayed set. These are not just laboratory curiosities; they are causes of rework, warranty claims, and shortened service life.
Preventive measures are straightforward but require attention: verify materials, test trial mixes under realistic conditions, control site water and temperature, and follow proven curing regimes. Doing so transforms theoretical chemical knowledge into practical reliability.
Emerging trends in hydration science

The industry is moving toward lower-carbon binders and more efficient hydration. Blended cements, calcined clays, and novel activators aim to reduce Portland cement usage while maintaining or improving the hydration products that deliver strength. Research into engineered nanoparticles and sulfate-tolerant formulations seeks to tailor microstructure at finer scales.
Internal curing technologies and smart admixtures that release water or alter hydration kinetics on demand are growing in interest. These innovations aim to reconcile the competing demands of lower w/c for strength and sufficient water for full reaction, especially in high-performance mixes. The result could be concretes that are both stronger and greener.
There is also renewed attention on in-situ monitoring: embedded sensors that track temperature, moisture, and even chemical evolution during curing provide live data to optimize processes. This kind of feedback loop was uncommon a decade ago but is increasingly practical and cost-effective.
Practical checklist for maximizing strength through hydration
Below is a concise list of actionable steps to ensure hydration produces the desired strength and durability.
- Design mixes with appropriate w/c for the target strength and exposure.
- Select SCMs and admixtures consistent with jobsite conditions and schedule.
- Control temperature during mixing and curing, especially for mass pours.
- Ensure continuous curing to keep moisture available for hydration.
- Use trial batches and maturity monitoring to validate early-age performance.
- Inspect and verify aggregate compatibility to avoid ASR and other reactions.
A compact reference table: common cement phases and hydration products
| Phase (clinker) | Typical role in hydration | Primary products |
|---|---|---|
| C3S (alite) | Fast reaction, contributes to early strength | C-S-H, portlandite (CH) |
| C2S (belite) | Slow reaction, contributes to long-term strength | C-S-H, portlandite (CH) |
| C3A (aluminate) | Reacts quickly with gypsum, controls setting | Ettringite, later monosulfate |
| C4AF (ferrite) | Lower contribution to strength, affects color and sulfate behavior | Various hydrated aluminoferrites |
Putting it all together: design, monitor, adapt
Concrete strength is not the result of a single ingredient or moment; it is the outcome of orchestrated chemistry, careful proportioning, and attentive curing. Each decision — w/c ratio, SCM choice, curing regime, admixture selection — nudges the hydration pathway toward a denser or more porous end-state. Good results come from understanding these levers and using them intentionally.
Monitoring and flexibility are important. Field conditions change, and mixtures that performed well in the lab may behave differently onsite. Track temperature and moisture, use maturity methods where appropriate, and be ready to adapt curing or placement procedures to preserve hydration. That practical responsiveness prevents small chemical missteps from becoming structural problems.
When the chemistry, materials, and construction practices align, hydration produces a remarkably durable, load-bearing material from humble ingredients. The process is intricate but predictable, and mastering it yields structures that meet both immediate load demands and decades of service.
Concrete’s strength is ultimately a story of transformation — water coaxing reactive minerals to rebuild themselves into a continuous, resilient matrix. That transformation is why our bridges stand, our buildings shelter us, and our infrastructure endures when chemistry and craft work together.